The term PGS refers to preimplantation genetic diagnosis. This is in effect checking the embryo for genetic diseases before replacing it into the uterus.
The most common reason for this is that one or both partners are known to carry a genetic trait usually associated with a severe genetic disease. Another typical example is a couple who has given birth to a child with such a severe genetic disease. Some such diseases are uniformly fatal, while others are associated with severe disability. In another situation, the couple may know that they are carriers for a genetic trait ahead of time by a screening process. The most common severe genetic diseases in the US include sickle cell anemia, cystic fibrosis, Tay Sachs disease, spinal muscular atrophy and Huntington’s disease.
This process begins with a standard IVF cycle. For full details of this, please go to the IVF section of the web site. Briefly, the ovaries are stimulated with medication. The eggs are harvested by ultrasound. Each egg is injected with a single sperm (ICSI). This is done to prevent the embryo from being covered with sperm DNA which can contaminate the embryo biopsy. The fertilized eggs (embryos) are then incubated for 3 days. Some embryos will naturally stop dividing. Others will be healthy and continue to divide. Healthy embryos which are at the 6 – 8 cell stage can then be biopsied. The biopsy technique involves removing carefully one cell and either fixing it to a slide or releasing its DNA for further analysis. The image below is of such a biopsy in progress.
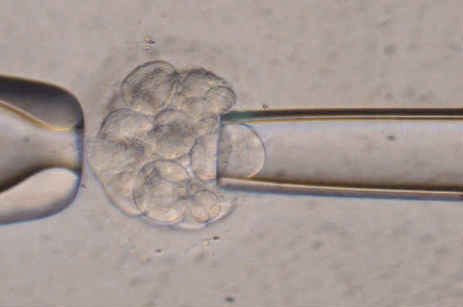
Biopsy in progress.
Typically the biopsy is done by the IVF program and the genetic material is sent off to a genetic lab which is frequently in another city. The lab will then try to get results within the next 48 hours. If the results are available in that time frame, we have an opportunity to do a fresh embryo transfer usually 5 or 6 days after egg retrieval. If the results cannot be obtained within this time frame, the embryos can be frozen until the results are known. Then a frozen embryo transfer can be performed. Some programs will routinely biopsy at the blastocyst stage and freeze all the embryos. Transfer would later occur in a frozen cycle.
PGS is highly complex. It requires 2 teams of lab staff, a physician and a genetics expert. It is surprising given its complexity that it only adds $4,000.00 to $5,000.00 to the cost of the typical IVF cycle.
It is not. It is a new technique and cannot test for all genetic defects at once. It can typically test for one at a time. It is too early to say it is 100% effective for testing for that one gene. Most recent studies show that it is more than 90% effective for testing for one genetic defect.
Yes, PGS can prevent miscarriage related to chromosomal errors such as trisomy and unbalanced translocation.
Some programs will allow this and others will not. In our program, we do allow it in cases of family balancing. To qualify for this, a couple must have 2 or more children of the same sex and wish to have a child of the opposite sex.
It typically takes 6 months to develop specific probes for the individual gene. Usually, blood has to be initially collected from the parents and tested. The probes are then developed.
The pregnancy rate will vary from 50% for young patients to less than 20% for patients in their forties.
Not always. In most cases, some of the embryos will be normal and available for transfer. In some cases, all of the embryos can be abnormal and therefor not suitable for transfer.